 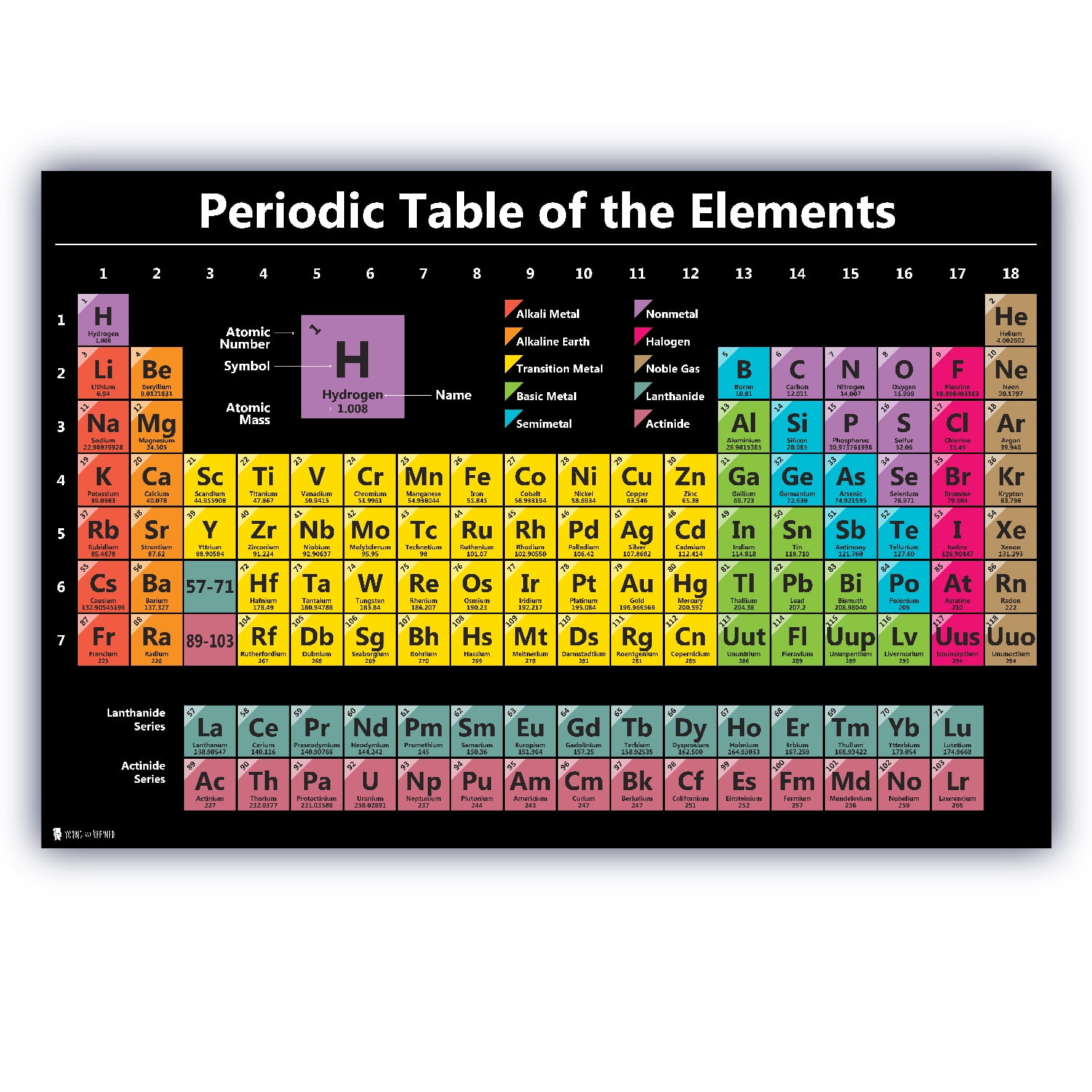
– Access at a full resolution of this Table as PDF (made available by King’s Center for Visualization in Science).īy virtue of its work in relation with the chemical elements, IUPAC can dispense a periodic table that is up-to-date. – Review the latest IPTEI element-by-element review including a chart of all known stable and radioactive isotopes for each element and examples practical applications of isotopic measurements and technologies – Explore the interactive version at /isotopes-matter (or see release) – Read “Atomic Weights: No Longer Constants of Nature”, Chem Int 33(2), 10–15 (2011), IUPAC Periodic Table of the Elements and Isotopes (IPTEI) for the Educational Community Coplen, Peter Mahaffy, Ian Mills, Roberto Marquardt, and more. Leigh, Sigurd Hofmann, Eric Scerri, Juris Meija, Norman E. See PAC (AOP ) for full details or visit Commission II.1 ĭownload the PDF version (letter size or A4) or A3 (PDF) or see earlier versionsĬheck out SPECIAL Chem Int Jan 2019 - International Year of the Periodic Table (IYPT) - with contributions by Jan Reedijk, Natalia Tarasova, G.J. For elements that lack isotopes with a characteristic isotopic abundance in natural terrestrial samples, the mass number of the nuclide with the longest confirmed half-life is listed between square brackets. However, even these small quantities are required for the body to function properly.The latest release of the Periodic Table (dated ) includes the most recent abridged standard atomic weight values released by the IUPAC Commission on Isotopic Abundances and Atomic Weights ( CIAAW), compiled as part of the 2021 Table of Standard Atomic Weights 2021. These last three metals are present in the body in very small quantities. Cobalt is a necessary component of vitamin B-12, a vital nutrient. Manganese is needed for the body to metabolize oxygen properly. Copper is also needed for several proteins to function properly in the body. Zinc is needed for the body’s immune system to function properly, as well as for protein synthesis and tissue and cell growth. Other transition metals have important functions in the body, despite being present in low amounts. It is the presence of this particular transition metal in your red blood cells that allows you to use the oxygen you inhale. Each hemoglobin molecule has four iron atoms, which act as binding sites for oxygen. The crucial atom in the hemoglobin protein is iron. Hemoglobin is a relatively large molecule, with a mass of about 65,000 u. Hemoglobin combines with oxygen and carbon dioxide, transporting these gases from one location to another in the body. The critical part of the red blood cell is a protein called hemoglobin. Without red blood cells, animal respiration as we know it would not exist. Red blood cells are cells that transport oxygen from the lungs to cells of the body and then transport carbon dioxide from the cells to the lungs. Iron is a transition metal and the chemistry of iron makes it a key component in the proper functioning of red blood cells. Because iron has relatively massive atoms, it would appear even lower on a list organized in terms of percent by atoms rather than percent by mass. The most abundant non-main group element is iron, at 0.006 percentage by mass. Most of the elemental composition of the human body consists of main group elements. To Your Health: Transition Metals in the Body Each group is located in a different part of the periodic table. Elements are either metals, nonmetals, or semimetals. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
